ATRX/Daxx Antibody Sampler Kit #95830
To Purchase # 95830
| Cat. # | Size | Qty. | Price |
|---|---|---|---|
| 95830T | 1 Kit 4 x 20 microliters | $410 |
- Product Includes
- Related Products
| Product Includes | Quantity | Applications | Reactivity | MW(kDa) | Isotype |
|---|---|---|---|---|---|
| ATRX (D1N2E) Rabbit mAb #14820 | 20 µl | WB, IP, ChIP | H | 280 | Rabbit IgG |
| Daxx (25C12) Rabbit mAb #4533 | 20 µl | WB, IF | H M R | 110 | Rabbit IgG |
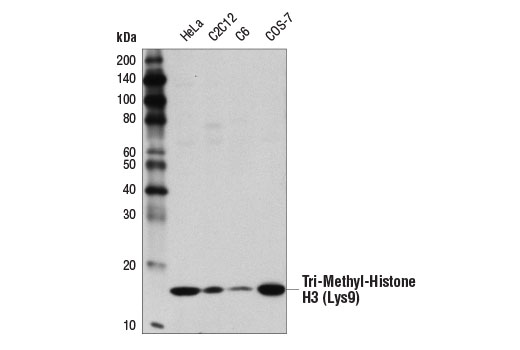
| Tri-Methyl-Histone H3 (Lys9) (D4W1U) Rabbit mAb #13969 | 20 µl | WB, IP, IF, F, ChIP | H M R Mk | 17 | Rabbit IgG |
| Histone H3 (D1H2) XP® Rabbit mAb #4499 | 20 µl | WB, IHC, IF, F | H M R Mk | 17 | Rabbit IgG |
| Anti-rabbit IgG, HRP-linked Antibody #7074 | 100 µl | WB | Rab | Goat |
Product Information
Kit Usage Information
Protocols
- 4499: Western Blotting, Immunohistochemistry (Paraffin), Immunofluorescence*, Flow
- 4533: Western Blotting, Immunofluorescence
- 7074: Western Blotting
- 13969: Western Blotting, Immunoprecipitation (Magnetic), Immunofluorescence*, Flow, ChIP Magnetic
- 14820: Western Blotting, Immunoprecipitation (Agarose), ChIP Magnetic
Product Description
Background
Daxx is a ubiquitously expressed protein that was originally identified through a yeast two-hybrid screen as an interactor with the cytoplasmic domain of Fas. It was found to enhance Fas-mediated apoptosis and activate the JNK pathway (17). However, additional studies have revealed that Daxx is actually a nuclear protein localizing to promyelocytic leukemia oncogenic domains (PODs) (18,19). Nuclear interactions have since been observed with CENP-C (20), Pax3 (22), DNA methyltransferase I (21) and chromatin-associated proteins, including histone deacetylase II, H2A, H2B, H3, H4, and Dek. Roles for Daxx have been suggested in transcriptional repression and cell cycle control. Loss of Daxx in mice leads to embryonic lethality with extensive developmental apoptosis, suggesting a role for Daxx directly or indirectly in suppressing cell death (22). Furthermore, inhibition of Daxx expression using RNAi has confirmed Daxx to be anti-apoptotic and to repress transcriptional activity of targets, including NF-κB and E2F-1 (23).
- Clynes, D. et al. (2013) Trends Biochem Sci 38, 461-6.
- Picketts, D.J. et al. (1996) Hum Mol Genet 5, 1899-907.
- Drané, P. et al. (2010) Genes Dev 24, 1253-65.
- Elsässer, S.J. et al. (2012) Nature 491, 560-5.
- Lewis, P.W. et al. (2010) Proc Natl Acad Sci U S A 107, 14075-80.
- Goldberg, A.D. et al. (2010) Cell 140, 678-91.
- Ritchie, K. et al. (2008) J Cell Biol 180, 315-24.
- De La Fuente, R. et al. (2004) Dev Biol 272, 1-14.
- Wong, L.H. et al. (2010) Genome Res 20, 351-60.
- Gibbons, R.J. et al. (2000) Nat Genet 24, 368-71.
- Gibbons, R.J. et al. (1995) Cell 80, 837-45.
- Gibbons, R.J. et al. (1995) Hum Mol Genet 4 Spec No, 1705-9.
- Heaphy, C.M. et al. (2011) Science 333, 425.
- Lovejoy, C.A. et al. (2012) PLoS Genet 8, e1002772.
- Schwartzentruber, J. et al. (2012) Nature 482, 226-31.
- Jiao, Y. et al. (2011) Science 331, 1199-203.
- Yang, X. et al. (1997) Cell 89, 1067-76.
- Torii, S. et al. (1999) EMBO J 18, 6037-49.
- Li, H. et al. (2000) Mol Cell Biol 20, 1784-96.
- Pluta, A.F. et al. (1998) J Cell Sci 111 (Pt 14), 2029-41.
- Michaelson, J.S. et al. (1999) Genes Dev 13, 1918-23.
- Hollenbach, A.D. et al. (1999) EMBO J 18, 3702-11.
- Suihko, M.L. and Stackebrandt, E. (2003) J Appl Microbiol 94, 25-34.
Pathways
Explore pathways related to this product.
Limited Uses
Except as otherwise expressly agreed in a writing signed by a legally authorized representative of CST, the following terms apply to Products provided by CST, its affiliates or its distributors. Any Customer's terms and conditions that are in addition to, or different from, those contained herein, unless separately accepted in writing by a legally authorized representative of CST, are rejected and are of no force or effect.
Products are labeled with For Research Use Only or a similar labeling statement and have not been approved, cleared, or licensed by the FDA or other regulatory foreign or domestic entity, for any purpose. Customer shall not use any Product for any diagnostic or therapeutic purpose, or otherwise in any manner that conflicts with its labeling statement. Products sold or licensed by CST are provided for Customer as the end-user and solely for research and development uses. Any use of Product for diagnostic, prophylactic or therapeutic purposes, or any purchase of Product for resale (alone or as a component) or other commercial purpose, requires a separate license from CST. Customer shall (a) not sell, license, loan, donate or otherwise transfer or make available any Product to any third party, whether alone or in combination with other materials, or use the Products to manufacture any commercial products, (b) not copy, modify, reverse engineer, decompile, disassemble or otherwise attempt to discover the underlying structure or technology of the Products, or use the Products for the purpose of developing any products or services that would compete with CST products or services, (c) not alter or remove from the Products any trademarks, trade names, logos, patent or copyright notices or markings, (d) use the Products solely in accordance with CST Product Terms of Sale and any applicable documentation, and (e) comply with any license, terms of service or similar agreement with respect to any third party products or services used by Customer in connection with the Products.